Bohr's Atomic Model
Rutherford, explain!
Accelerating charges ought to loose energy in the form of γ rays.
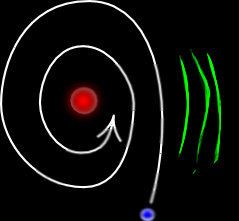
What, then, makes the atom stable?
Rutherford, explain!
Even if the atom did loose energy, it should have been continuous...
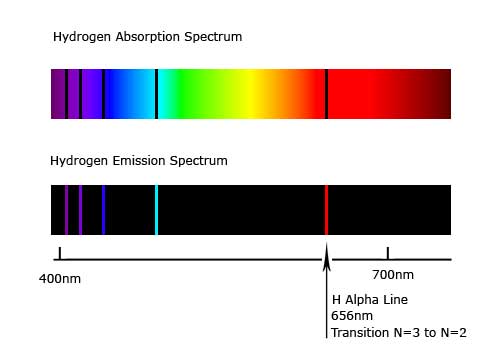 ... which it wasn't.
... which it wasn't.
We need a better theory.
Bohr's Postulates
- Electrons in atoms orbit the nucleus in circular orbits.
- There exists discrete stationary orbits in which the electrons orbit stably without radiating.
- It turns out that the angular momentum of these orbits are multiples of ħ
- When electrons jump from orbit to orbit, they emit/lose energy according to Plank radiation.
Radius
-
Equating Centrapetal Force and Electric Force,
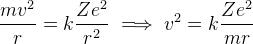
-
From Bohr's second postulate,
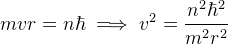
-
Therefore,

-
And we have the radius,
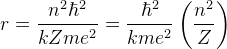
Velocity
Plugging in the radius gives the velocity,
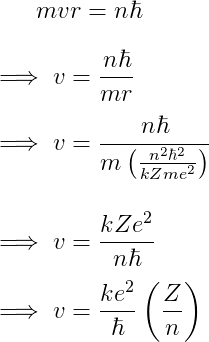
Energy
-
Kinetic Energy:
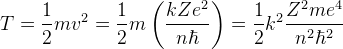
-
Potential Energy:
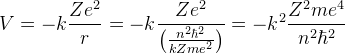
-
Total Energy:
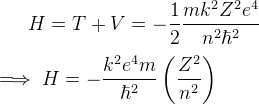
Matter Waves
- Matter does behave like a wave, in some sense.
- deBroglie hypothesised that the associated wavelength of a particle is given by λ = h/p
Matter Waves
Seen carefully enough, electrons produce diffraction patterns.
deBroglie's Justification
- Bohr's orbitals are stationary states where the wave function does not evolve.
- Consider an electron wavicle of n wavelengths wrapped around a Bohr orbit.
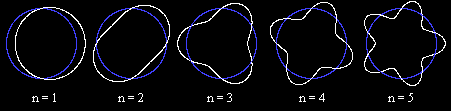
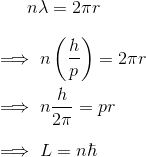
Not the end...
- Still too many spectral lines
- What if there is more than one electron?
- Too classical: Newtonian mechanics is hardly a good way to describe phenomenon at the quantum level.
Location Sharing

Thank You!
/